Eukaryotes use different families of chaperone Molecules of a second chaperone, GroES, then form a Of the chaperone GroEL form a hollow chamber around proteins that are in Proteins surround a protein during the folding process, sequestering the Inappropriate associations with unintended folding partners. Cells therefore rely on so-called chaperone Nearby proteins can interfere with proper folding and cause large Interacting with a partially folded protein. Is a crowded place, filled with many other macromolecules capable of Their final shapes without any energy input. Theory, once their constituent amino acids are strung together, proteins With this method, solid crystals of purified protein are placed in an X-ray beam, and the pattern of deflected X rays is used to predict the positions of the thousands of atoms within the protein crystal. The most common method used to study protein structures is X-ray crystallography. So, scientists must use indirect methods to figure out what they look like and how they are folded. Rather, the atoms within these proteins remain capable of making small movements.Įven though proteins are considered macromolecules, they are too small to visualize, even with a microscope. It is important to note, however, that fully folded proteins are not frozen into shape. In contrast, the proteins that are inserted into the cell membranes display some hydrophobic chemical groups on their surface, specifically in those regions where the protein surface is exposed to membrane lipids. For example, the proteins that are dissolved in the cell cytoplasm have hydrophilic (water-loving) chemical groups on their surfaces, whereas their hydrophobic (water-averse) elements tend to be tucked inside. In addition, chemical forces between a protein and its immediate environment contribute to protein shape and stability. Folded proteins are stabilized by thousands of noncovalent bonds between amino acids. As proteins fold, they test a variety of conformations before reaching their final form, which is unique and compact. The final shape adopted by a newly synthesized protein is typically the most energetically favorable one. Finally, the quaternary structure of a protein refers to those macromolecules with multiple polypeptide chains or subunits. The ensemble of formations and folds in a single linear chain of amino acids - sometimes called a polypeptide - constitutes the tertiary structure of a protein. Most proteins contain multiple helices and sheets, in addition to other less common patterns (Figure 2). Known as alpha helices and beta sheets, these stable folding patterns make up the secondary structure of a protein. Hydrogen bonding between amino groups and carboxyl groups in neighboring regions of the protein chain sometimes causes certain patterns of folding to occur. 
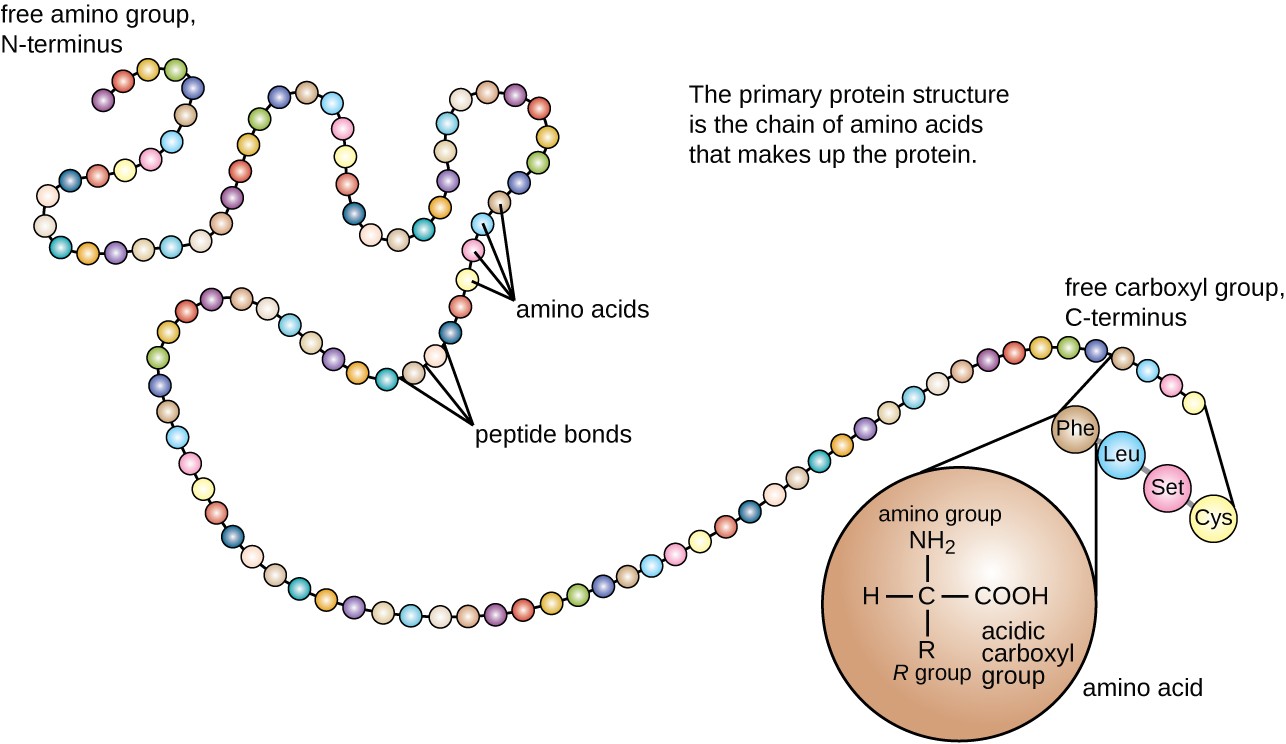
The primary structure of a protein - its amino acid sequence - drives the folding and intramolecular bonding of the linear amino acid chain, which ultimately determines the protein's unique three-dimensional shape. © 2010 Nature Education All rights reserved. Because of side chain interactions, the sequence and location of amino acids in a particular protein guides where the bends and folds occur in that protein (Figure 1). In fact, cysteines are the only amino acids capable of forming covalent bonds, which they do with their particular side chains. The vast majority of bonds formed by these side chains are noncovalent. Hydrophobic side chains interact with each other via weak van der Waals interactions. Charged amino acid side chains can form ionic bonds, and polar amino acids are capable of forming hydrogen bonds. The chemistry of amino acid side chains is critical to protein structure because these side chains can bond with one another to hold a length of protein in a certain shape or conformation. Several other amino acids have side chains with positive or negative charges, while others have polar but uncharged side chains. The largest group of amino acids have nonpolar side chains. The side chains of amino acids have different chemistries. Proteins are built from a set of only twenty amino acids, each of which has a unique side chain. The linear sequence of amino acids within a protein is considered the primary structure of the protein. 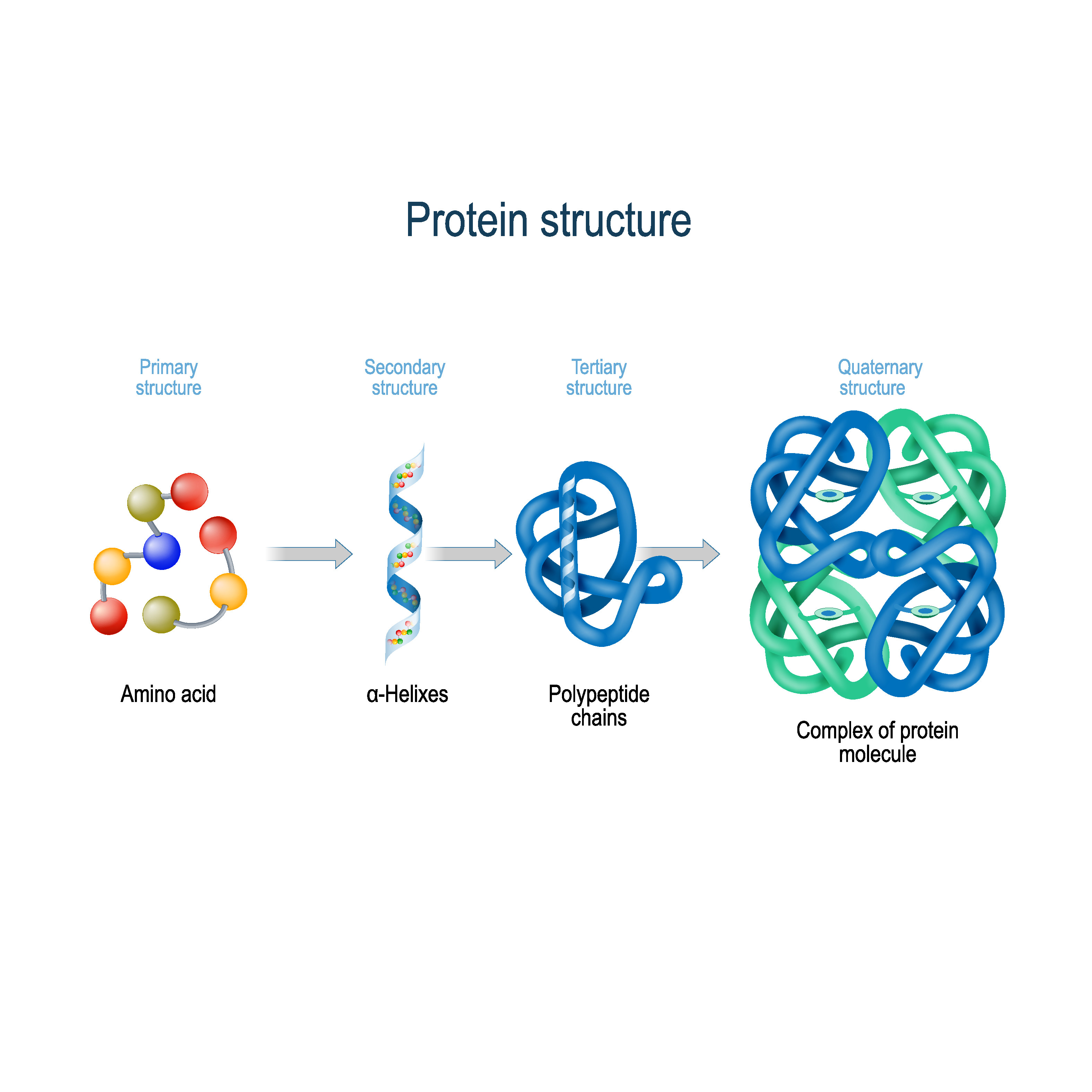
Peptide bonds are formed by a biochemical reaction that extracts a water molecule as it joins the amino group of one amino acid to the carboxyl group of a neighboring amino acid. Within a protein, multiple amino acids are linked together by peptide bonds, thereby forming a long chain. The building blocks of proteins are amino acids, which are small organic molecules that consist of an alpha (central) carbon atom linked to an amino group, a carboxyl group, a hydrogen atom, and a variable component called a side chain (see below).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
